|
|
|
|
|
|||||||||||
|
Electronic Flora of South Australia Species Fact Sheet
Phylum Rhodophyta – Order Ceramiales – Family Ceramiaceae – Tribe Ceramieae
Selected citations: De Toni 1903: 1491. Guiler 1952: 98. Lucas 1909: 53; 1929a: 26. Lucas & Perrin 1947: 369. Womersley 1978: 224, figs 2D, 10.
Synonyms
C. torulosum J. Agardh 1876: 99; 1894: 47. De Toni 1903: 1490. Guiler 1952: 99. Lucas 1909: 53; 1929a: 26. Sonder 1881: 12. (NON C. torulosum Roth 1806: 125 = Lomentaria articulata (Hudson) Lyngbye.)
C. nodiferum sensu J. Agardh 1894: 31. Lucas 1909: 53. Lucas & Perrin 1947: 369. Tisdall 1898: 503. Wilson 1892: 185. (NON J. Agardh 1876: 99, from the Chatham Is.)
C. apiculatum sensu Wilson 1892: 184. Tisdall 1898: 503. (NON J. Agardh 1876: 105.)
? C. stichidiosum sensu Lucas 1909: 53; 1929a 26. Guiler 1952: 98. Sonder 1881: 13. (NON J. Agardh 1876: 105.)
? C. nodiferum sensu Lucas 1909: 53. Lucas & Perrin 1947: 369. Tisdall 1898: 503. Wilson 1892: 185.
Thallus (P1. 2, fig. 4; Fig. 183A, B) medium to dark red, 5–20 (–30) cm high with a single or several erect axes (or occasionally a slight prostrate base) producing several long lateral branches, usually complanately branched (Fig. 183B) and regularly alternate in younger parts but below often more irregularly branched, often with small, proliferous, often fertile branchlets along the lower main branches. Attachment by a tuft of uniseriate-celled rhizoids; epilithic. Structure. Branches 0.5–1.5 mm in diameter below, decreasing gradually to 150–250 µm in diameter just below the relatively straight, markedly tapering apices; proliferous branchlets on older branches 120–180 µm in diameter, often basally constricted. Axial cells L/D about 1 above, extending to L/D 2 in older branches, becoming corticated except for a narrow, annular, internodal space (occasionally almost as long as the nodes in upper parts) present throughout the thallus (Figs 182D, 183C), but with the cortication usually closing completely on older branches and extending as the axial cells enlarge. Periaxial cells usually 8, each cutting off a regular pattern of 2 cells acropetally and 2 (rarely 3) basipetally (Fig. 182D), these continuing as corticating filaments usually each with two divisions, occasionally one; divisions normally synchronous, producing a straight edge to the cortication (except where axial cells have elongated relative to cortical development), with smaller terminal cells on the acropetal filaments than on the basipetal ones (Fig. 182D), ultimately often with further production of small cells closing the internodal space completely. Outer cortex produced from the periaxial cells (Figs 182D, 183C) and some of the larger inner cortical cells (which elongate to L/D up to 5), often with well developed rosettes over these cells, and on older parts a further cortical layer of small cells may be cut off, forming a distinctly thicker cortex over the central part of the nodal cortication; in some specimens, scattered small outer cortical cells become gland-like. Rhodoplasts discoid in very small cells, ribbon like and longitudinal in axial cells.
Reproduction: Gametophytes dioecious. Carposporophytes (Fig. 183D) near the ends of short lateral branchlets, globular, 200–400 µm across, with 2–4 (–5) short, relatively stout, involucral branches; carposporangia ovoid, 22–30 µm in diameter. Spermatangial patches covering several segments, largely in the axils of upper branches, sometimes surrounding small lateral branches.
Tetrasporangia (Fig. 183E) formed mainly in stichidiose branch ends or proliferous laterals which are basally constricted, derived from periaxial cells and forming a ring of 8–12 lying within and bulging the acropetal cortical filaments (Fig. 183E), thus producing a torulose appearance to the branch; tetrasporangia subspherical to slightly ovoid, 30–50 µm in diameter, decussately divided.
Lectotype from Tamar R. mouth, Tasmania (Oakden); in Herb. Agardh, LD, 21757.
Selected specimens: Head of the Great Australian Bight, S. Aust., drift (Parsons, 4.xi.1968; AD, A34180). Price I., off Point Avoid, S. Aust., 21m deep (Broaden, 6.i.1989; AD, A59971). Investigator Strait, S. Aust., 31m deep (Watson, 24.i.1971: AD, A38371). Picnic Point, American R. inlet, Kangaroo I., S. Aust., 2–3m deep (Womersley, 23.i.1948; AD, A8186). Robe, S. Aust., drift (Wollaston, 17.iii.1956; AD, A20391). Port MacDonnell, S. Aust., drift (Womersley, 26.ix.1992; AD, A62990). Port Phillip Heads, Vic., 22m deep (Watson, 29.iii.1985; AD, A57044-"Marine Algae of Southern Australia" No. 314). Low Head, Tas. (Perrin, Sept. 1948; AD, A9020). Cloudy Lagoon, SE Tas., 1–3m deep (Edgar, 12.ii.1995; AD, A64234).
Distribution: Head of the Great Australian Bight, S. Aust, to Western Port, Vic., and around Tasmania. This appears to be usually a deep water species.
Taxonomic notes: C. excellens is usually well characterised by its normally complanate form and the presence throughout much of the thallus of the clearly defined, narrow and straight edged, annular internodal space; this is usually longest nearer the apices, but is rarely more than one fifth as long as the nodal cortication (except in specimens with enlarged axial cells), and in the older branches the cortex often closes completely.
C. excellens is closely related to C. lenticulare, but differs in habit, greater dimensions, and in details of the nodal cortication, especially in the uniform presence of the narrow, annular internodal space compared to the lenticular internodal space in C. lenticulare. Whereas the axial cells of C. lenticulare remain isodiametric throughout, in larger branches of C. excellens they elongate to L/D about 2 and the cortex extends to maintain complete cover of these cells; the latter also develops a considerably greater outer cortex.
C. excellens is also closely related to C. tasmanicum, but differs in having complanate branching (at least in younger parts) and in more regular nodal cortication with the narrow, annular internodal space; C. excellens usually also has a thicker cortex and well defined rosettes in the central part of the nodal cortication. Some apparent intermediates between these two species do occur.
References:
AGARDH, J.G. (1876). Species Genera et Ordines Algarum. Vol. 3, Part 1- Epicrisis systematic Floridearum, pp. i-vii, 1–724. (Weigel: Leipzig.)
DE TONI, G.B. (1903). Sylloge Algarum omnium hucusque Cognitarum. Vol. 4. Florideae. Sect. 3, pp. 775–1521 + 1523–1525. (Padua.)
GUILER, E.R. (1952). The marine algae of Tasmania. Checklist with localities. Pap. Proc. R. Soc. Tasmania 86, 71–106.
LUCAS, A.H.S. & PERRIN, F. (1947). The Seaweeds of South Australia. Part 2. The Red Seaweeds. (Govt Printer: Adelaide.)
LUCAS, A.H.S. (1909). Revised list of the Fucoideae and Florideae of Australia. Proc. Linn. Soc. N.S.W. 34, 9–60.
LUCAS, A.H.S. (1929a). The marine algae of Tasmania. Pap. Proc. R. Soc. Tasm. 1928, 6–27.
ROTH, A.G. (1806). Catalecta Botanica. Vol. 3. (Lipsiae.)
SONDER, O.W. (1881). In Mueller, F., Fragmenta Phytographiae Australiae. Supplementum ad volumen undecinum: Algae Australianae hactenus cognitae, pp. 1–42, 105–107. (Melbourne.)
TISDALL, H.T. (1898). The algae of Victoria. Rep. 7th Meet. Aust. Ass. Adv. Sci., Sydney, 1898, pp. 493–516.
WILSON, J.B. (1892). Catalogue of algae collected at or near Port Phillip Heads and Western Port. Proc. R. Soc. Vict. 4, 157–190.
WOMERSLEY, H.B.S. (1978). Southern Australian species of Ceramium Roth (Rhodophyta). Aust. J. Mar. Freshw. Res. 29, 205–257.
The Marine Benthic Flora of Southern Australia Part IIIC complete list of references.
Publication:
Womersley, H.B.S. (24 December, 1998)
The Marine Benthic Flora of Southern Australia
Rhodophyta. Part IIIC. Ceramiales – Ceramiaceae, Dasyaceae
©State Herbarium of South Australia, Government of South Australia
Illustrations in Womersley Part IIIA, 1998: PLATE 2 fig. 4; FIGS 182D, 183.

Plate 2 enlarge
PLATE 2 fig. 1. Anotrichium elongatum at Robe, S. Aust. (AD, A63206).
fig. 2. Euptilota articulata at Rottnest I., W.Aust. (MURU, JH727). Photo: J.M. Huisman.
fig. 3. Ceramium rubrum at Cloudy Lagoon, SE Tas. (AD, A64230). Photo: G. Edgar.
fig. 4. Ceramium excellens at Cloudy Lagoon, SE Tas. (AD, A64234). Photo: G. Edgar.
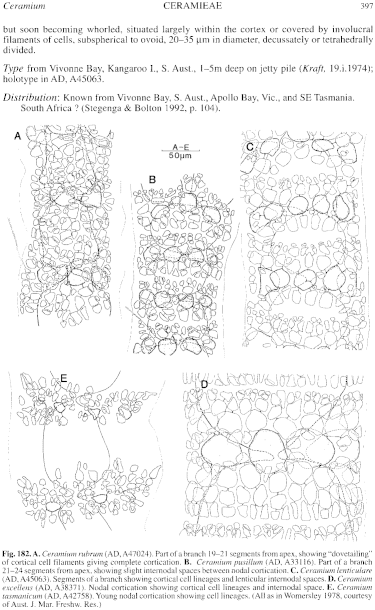
Figure 182 enlarge
Fig. 182. A. Ceramium rubrum (AD, A47024). Part of a branch 19-21 segments from apex, showing "dovetailing" of cortical cell filaments giving complete cortication. B. Ceramium pusillum (AD, A33116). Part of a branch 21-24 segments from apex, showing slight internodal spaces between nodal cortication. C. Ceramium lenticulare (AD, A45063). Segments of a branch showing cortical cell lineages and lenticular internodal spaces. D. Ceramium excellens (AD, A38371). Nodal cortication showing cortical cell lineages and internodal space. E. Ceramium tasmanicum (AD, A42758). Young nodal cortication showing cell lineages. (All as in Womersley 1978, courtesy of Aust. J. Mar. Freshw. Res.)

Figure 183 enlarge
Fig. 183. Ceramium excellens (A, C–E, AD, A38371; B, AD, A9020). A. Habit of deep water plant. B. Habit of well grown shallow water plant. C. Branch showing cortication, well defined annular internodal spaces, and rosette formation around periaxial cells. D. Carposporophyte with involucral branches. E. Branch with whorled tetrasporangia. (All as in Womersley 1978, courtesy of Aust. J. Mar. Freshw. Res.)

|
Email Contact: State Herbarium of South Australia |

|