Family: Fabaceae
Acacia saligna

Citation:
H. Wendland, Comment. Acac. 4, 26 (1820). Mimosa saligna Labill., Nov. Holl. Plant. Specim. 2:86 t.235 (1807).
Derivation: salignus (Latin)--of willow, used in the sense of willow-like. Synonymy: Acacia cyanophylla Lindl., Edward's Bot. Reg. 25 Misc. 45 (1839). Racosperma salignum (Labill.)Pedley, Austrobaileya 2:355 (1987)., Acacia bracteata, Acacia lindleyi, Mimosa saligna Common name: Golden wreath wattle
Description:
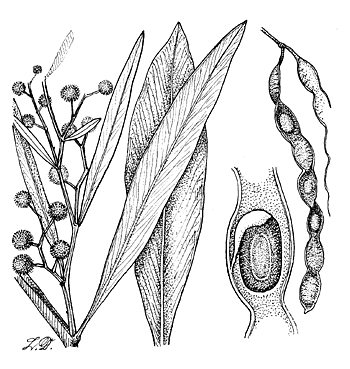
Dense, bushy shrubs or small trees 3-8 m high; branchlets sometimes pendulous, flattened towards the apex but soon becoming almost terete; trunk usually short, dividing just above the ground level into a few main stems; bark smooth, grey to reddish brown, often producing suckers. Phyllodes variable, linear to lanceolate, 8-25 cm long, 2-50 mm broad, straight or falcate, tapering gradually towards the base and apex, glabrous, often glaucous when young, mid-vein prominent with fine but distinct lateral veins; glands basal. Inflorescences axillary racemes, shorter than phyllodes (rarely reduced to a solitary head), axis slightly flexuose, glabrous; flower-heads globular, deep yellow to almost orange, 25-75-flowered; peduncles slender, glabrous, 5-20 mm long; flowers 5-merous. Legumes linear, 5-14 cm long, 5-6 mm broad, straight or slightly curved, surface slightly undulate, glabrous, brown, margins thickened, yellowish, slightly constricted. Seeds longitudinal in legume, obloid; funicle short, thickening into an aril.
|
|
Distribution:
|
Due to its suckering habit plants once established may persist for some time even when the original plant has died. It is not always possible to decide if these suckering remnants should be considered as naturalised. Better notes with collections are necessary. Questionable specimens have come from Eyre Peninsula, Yorke Peninsula, Southern Lofty, Murray, Kangaroo Island and South-Eastern regions. Soils: Calcareous sands, leached sands with a hardpan. Rainfall approx. 300-700 mm. Endemic to W.Aust., introduced into Qld, N.S.W. and Vic.
S.Aust.: EP, MU, YP, SL, KI, SE.
|
Flowering time: August — October. |
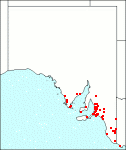
SA Distribution Map based
on current data relating to
specimens held in the
State Herbarium of South Australia
|
Biology:
No text
Taxonomic notes:
The application of the names A. saligna and A. cyanophylla has long been confused. Maslin (1974) studied the material available and considered A. cyanophylla to be a synonym of A. saligna. Maslin describes the species in detail, discusses variability which is considerable in W.Aust. and is evident in the plate of leaf sizes and shapes. A map of the natural distribution is given. Maslin considers that A. saligna is closely related to A. pycnantha and A. leiophylla. The last two rarely, if ever sucker, a point which is not made by Maslin, and it is the suckering habit which enables original plantings in S. Australia to persist and spread when the parent plant is destroyed.
Majer (1978) discussed the possible protective function of extra-floral nectaries. The activity of the glands on 20 marked phyllodes on forty plants along roadsides near Yalorup, W.Aust. was followed. Later 20 trees at Manning were added to the study and ants were excluded from some trees. Herbivore damage, gland and insect activity were all followed for about 12 months. A diverse and abundant range of damaging herbivores was found. Aggressive ants were present, the locality and time of gland activity supported any protective effect of ants. Glands were most active in spring and autumn when the threat of herbivory is greatest but the results were not conclusive. The possible role of the extrafloral nectaries in attracting bird pollinators (cf. A. pycnantha) was not discussed.
The natural insect enemies of A. saligna and A. cyclops in Western Australia have been studied by Van den Berg (1980a, b, c). Larvae of 36 species of Lepidoptera (moths and butterflies) were found on A. saligna of which 14 also occurred on A. cyclops. Those damaging the phyllodes were the most common. Adults or larvae of 55 species of Coleoptera (beetles and weevils) were found on A. saligna of which only 11 species were also found on A. cyclops. Adults and/or nymphs of 40 species of Hemiptera (Cicadas, Plant Hoppers, Plant Lice, Scale Insects and Bugs) were found on A. saligna. Those feeding on sap and twigs were most abundant. The study was done to establish the importance of natural enemies with a view to biological control of these wattles in South Africa.
Nakos (1977) found that the ability to fix nitrogen was greatly reduced by drought, waterlogging, shading or defoliation (as A. cyanophylla).
For the results of seven pre-sowing treatments on germination (as A. cyanophylla) see Aveyard (1968).
The nutritional value of A. cyanophylla seeds was studied by Ramadan (1957) on material grown in Cyprus. The gross protein value was about the same as ground nut meal. Growth of chickens with protein supplied by Acacia seed was inferior to protein from casein. High levels of seed in the diet (> 34%) depressed chicken growth.
Acacia saligna has become established in South Africa, Ross (1975) where it is now common in the Cape Province. It was introduced about 1870 to stabilize dune sands in which it was successful but has spread extensively and is considered a menace as it invades and displaces indigenous vegetation.
Holmes (1989) presents an account of the decay rates in buried seed populations of different densities. Loss rates were high and up to 45% of the seeds tested either germinated or rotted by the end of the first year. There was no significant effect of density. The species is also locally established in southern Europe (as A. cyanophylla).
A recent study in South Africa, Musil & Midgley (1990) has shown that Acacia infestations are significant in changing the chemical status of soils. The changes are due to the greatly increased litter-fall mass under Acacia compared with the native fynbos heathy vegetation. The influence was greater than that of fire or seasonal climatic variation.
Seed removal rates in the litter of Acacia infested vegetation in the (fynbos) heathy areas of South Africa have been studied recently, Holmes (1990). There was a loss of 50% of seedfall in dense A. saligna and of 80-95% in dense A. cyclops stands. Ants were most active in removing seed and rodents less significant. The results indicate that indigenous ants have played a crucial role in maintaining and accumulating Acacia seed banks and thus may have facilitated the development of dense Acacia stands.
A. saligna is listed as being cultivated in the Adelaide Botanic Gardens in 1859, 1871, 1878 and a herbarium specimen from 1895 survives. It was not until the 1960s that collections came in from many areas. As the species suckers many planted specimens now form small colonies and it is difficult to assess the extent to which it is truly naturalised.
In cultivation in the Waite Arboretum 10 trees of A. saligna lived from 10 to 20 years averaging 14.9 years.
The leaves of A. saligna can be used to dye wool a lemon yellow colour using an alum mordant, Martin (1974).
Hall (1976) deals with A. saligna as No. 4 in a limited series on Australian Acacias. The geographical distribution and ecology, botanical features and utilisation are covered and a brief bibliography is provided.
Cultivation:
Well known in cultivation and a valuable hardy ornamental or small shade tree. It is suitable for soil stabilisation and does well in sand in near coastal areas. Fast growth rate.
Author:
Not yet available
References:
Source:
|

